30 Jun 2022

Every one of us knows how brutal cancer can be.
It is a mysterious and deadly disease: cancer happens when a body’s own cells divide uncontrollably and spread into surrounding tissues. This rapid development of abnormal cells has the ability to infiltrate and destroy normal body tissue. Today, cancer is the second-leading cause of death in the world.
And when people think of a ‘cure for cancer’, they may perhaps picture chemotherapy: a treatment that uses powerful chemicals to kill fast-growing cells in your body. But what many may not be fully aware of is the indiscriminate and off-target killing of cells that chemotherapy unfortunately performs.
Incidentally, that’s why so many cancer patients often lose their hair during chemotherapy. While chemotherapy is powerful, it's impossible to avoid causing some damage to other cells and tissues in the body. And when patients receive chemotherapy, some get sick - sometimes very sick.
Next-Generation Therapy Brings Hope to Patients
Enter Tessa Therapeutics, a clinical-stage biotechnology company developing novel next-generation, targeted cell therapies for cancer.
In layman terms, it is working towards creating safe and effective therapies for patients with advanced cancers, who have no treatment alternatives.
“Tessa Therapeutics began ten years ago in 2012. When I joined them seven years ago, the company only had a staff of about 30 people,” CTO and acting CEO, John Ng, says. Today, the company employs over 160 members of staff and is nestled in the south of Singapore, at 138 Depot Road with their own manufacturing facility.

John Ng, CTO and acting CEO of Tessa Therapeutics. (Photo: Tessa Therapeutics)
For the uninitiated, CD30 is a type of protein marker found on several types of lymphoma or blood cancer cells. It is homogeneously expressed in Hodgkin Lymphoma and many other subsets of non-Hodgkin Lymphomas.

These patients represent a significant unmet medical need.
CAR-T Approach Shows Promising Results in Early Trials
Tessa’s approach to curing this deadly cancer is through a targeted approach called CAR-T (Chimeric Antigen Receptor) therapy. Their recent work has demonstrated excellent safety and efficacy in early clinical trials, and many are hopeful that this will usher in a new age of hope for cancer patients.
After seven years of research and work, Tessa has begun advancing this therapy to a pivotal trial; the final leg of the clinical trials it must go through.
(Photo: Tessa Therapeutics’ website)
It goes without saying, then, that John is hopeful: “The current complete response rate for the oncology drugs and treatments available on the market for last line Hodgkin Lymphoma patients sits at around 20-30%. Our therapy has shown a 57% complete response rate, and a 70% objective response rate, which includes both partial and complete responses.”
A partial response is a decrease in the amount of cancer in the body, while a complete response is the disappearance of all signs of cancer in the body. The higher the complete and partial response rate, the better for the cancer patient.
Tessa reached this level of success due to their extensive and ground-breaking research on CAR-T cell therapy.
How the CAR-T Cell Therapy Works
Essentially, CAR-T therapy uses one’s one immune cells, called T-cells, to fight cancer cells by genetically altering them in a laboratory. T-cells have proteins called receptors that look for proteins on foreign substances, called antigens, to lock onto and trigger other parts of the immune system to destroy it. Cancer cells also have antigens but just like a lock and key, it requires the right T-cell receptors to be able to attach onto it to destroy the cancer cells. CAR-T adds a gene to the receptor so that it becomes the right “key” to fit into the antigen “lock”.
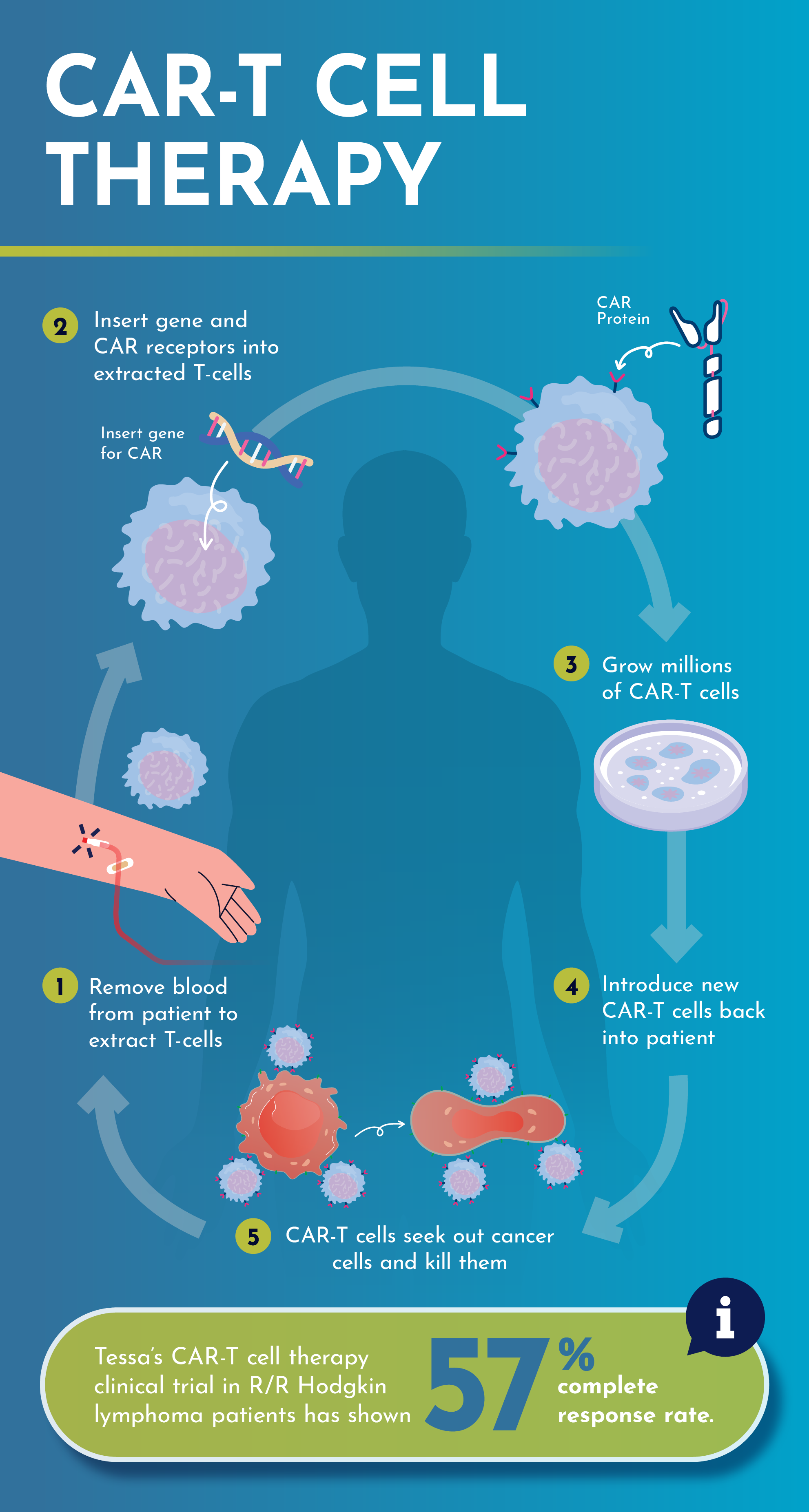
However, the challenge for this form of targeted personalised cell therapy is the difficulty to scale and make the drug more accessible.
“This personalised CAR-T cell therapy means that each individual patient will receive treatment that has been personalised to their own immune system. Thus, the treatment for one patient would not be able to work for another,” he explains.
Making Cancer Treatment Accessible for All
In order to make a similar form of this therapy more accessible to the masses, Tessa is also focusing on off-the-shelf allogeneic cell therapy. This off-the-shelf medicine presents significant advantages - in terms of the response rate, accessibility as well as cost.
This unique and potentially transformational off-the-shelf allogeneic therapy- called CD30-CAR EBVST - is capable of targeting a broad range of cancers, and is the next frontier in cancer treatment.
Tessa aims to be one of the first companies to get these two therapy treatments approved.

Medicine produced by Tessa are living cells, hence they are stored in the automated cryogenic storage equipment under -150 °C before shipping to the hospital sites. (Photo: Tessa Therapeutics)
All Systems Go for Tessa Therapeutics
As of mid-2022, the young biotech start-up had just completed another round of fund-raising. The $126 million USD they raised will help the ambitious company in their mission to develop and produce cancer cell therapy for the public.
And thanks to some smart planning, these products will be manufactured in their own state-of-the-art manufacturing facility, where their headquarters is located.

Tessa’s headquarters at 138 Depot Road
The Tessa team recounted the many considerations they had when building their facilities.
Firstly: “An accessible and central location helps with employee attraction and retention,” John reveals. Tessa makes a huge investment with each employee they hire. The exacting standards of their work often means that new members undergo six to nine months of training. Should an employee choose to leave prematurely, it would represent a loss to the company.
“Our headquarters at Depot Road is central, near parks, and near residential areas. Most of the other bio-research and pharmaceutical companies are located in Tuas,” John adds. Indeed, Tessa Therapeutics is located 15 minutes from bustling malls in Harbourfront and Chinatown, and is a short walk from large swaths of greenery.
Secondly, and most importantly: “The facilities matched what our operations required,” John explains.
One example is the building's ability to support the operation of cleanrooms, which is critical to the production of Tessa’s therapy.
Cleanrooms must meet stringent performance standards, and in order to maintain such a high-level of cleanliness, the facility required several HEPA (High Efficiency Particulate Air) filtration systems that deliver air changes of up to 60 times per hour.
Another facility that was built from the ground up is their fully functional quality-control lab. “We do our in-house testing of products and their functionality in this room. The lab ensures both precision and accuracy of patient sample results as well as the rapid release of the medicine after quality-control checks,” John says.

Tessa does in-house testing of products within the quality-control lab. (Photo: Tessa Therapeutics)
“Singapore is a good and accessible location within Asia, with institutional life sciences investors and investors who want to do good and give back to society by helping to develop life-saving technology,” John reveals.
Shaping the Future of Healthcare
The many state-of-the-art, in-house capabilities of Tessa Therapeutics means that they are ushering in a new age of biotechnology in Singapore.

(Photo: Tessa Therapeutics’ website)
John’s hope for the future is for the entire biotech industry working in tandem. Tessa’s facilities, which could one day be utilised by future clinical stage biotechnology companies, helps create a flourishing ecosystem for the industry.
By pioneering game-changing healthcare technologies, Tessa Therapeutics will not only shape the future of healthcare - but shape the lives of hundreds of thousands of cancer patients, and their families.
Of his journey so far with Tessa Therapeutics, John shares:
We find ourselves in a very privileged position, with the potential to save lives.
John Ng, CTO of Tessa Therapeutics
Indeed, he lets us in on a small secret: many employees of the company gave up their past well-paying positions in the bio-pharma sector in order to work with Tessa Therapeutics in cell and gene therapy as they view that as one of the most promising modality for the future of cancer treatments.
More than just a passion project, the work at Tessa Therapeutics is one that, at its core, has the potential to save lives.
Submit your space enquiry
































